Estimated reading time: 11 minutes
Plant virus-vector nematodes are soil-dwelling, plant-feeding nematodes capable of transmitting plant viruses while feeding on roots or underground plant parts. Although only a small number of nematode species have this ability, their impact on crops such as potatoes, grapevines and fruit trees can be severe and persistent. Living below ground, and being microscopic these nematodes often escape notice, allowing viruses to remain in fields for many years (Girgan et al., in CHIPS September/October 2025). Recent findings linked to Pepper ringspot virus (PepRSV) in South Africa highlight the importance of understanding these hidden vectors and the role they play in disease spread.
A brief history of nematode-transmitted plant viruses
Only two of the 12 plant-feeding nematode families reported from South Africa are known to transmit plant viruses. The family Longidoridae transmit nepoviruses, while the family Trichodoridae transmit tobraviruses. Within the Longidoridae, virus transmission has been reported for species of the genera Xiphinema, Longidorus and Paralongidorus, while tobraviruses are transmitted by species of the genera Nanidorus, Paratrichodorus and Trichodorus. The first evidence of virus transmission by a plant-feeding nematode was reported in 1958, when Hewitt and co-authors in California demonstrated that Xiphinema index transmits Grapevine fanleaf virus. This discovery laid the foundation for understanding nematode-transmitted viruses globally, including in South Africa, where grapevine fanleaf virus has been reported for decades and its nematode vector is subject to regulatory control. Two years later Sol and co-authors demonstrated that Paratrichodorus pachydermus could transmit the tobacco rattle virus and in 1961 Walkinshaw and co-authors reported that Nanidorus minor is also a vector of Tobacco rattle virus, that causes corky ringspot on potato.
How nematodes transmit viruses
Upon feeding on viruliferous plants, the transformed sap passes through the lumen of the stylet and oesophagus. The virus particles attached to specific sites, in the trichodorids with the onchiostyle and cuticle lining of the oesophagus and in the longidorids the viruses are retained to sites on the oesophagus (Xiphinema) whereas as with Longidorus the virus particles usually attach in the lumen of the anterior part of the ondonstylet. All the life stages of the nematodes can acquire virus particles. The virus particles retained by nematodes do not pass through moulting or embryogenesis and are lost with shedding of the internal cuticle of nematode. Interestingly, when a viruliferous nematode feeds on a host, only a small portion of the retained virus particles are released and are thus available for future transmission. Pepper ringspot virus and Tobacco rattle virus are highly stable viruses that can persist in a field for several years, sometimes even decades, particularly when protected within their nematode vectors or in host weeds.
Long-term field studies on Tobacco rattle virus have shown that the virus can persist in soil for extended periods, often well beyond a single crop rotation cycle. In several European studies, TRV remained detectable and capable of infecting potatoes after five to ten years without a susceptible host crop. Notably, field trials in Scotland demonstrated that even after five consecutive years of crop rotation with non-host plants, including Medicago species that do not support populations of Paratrichodorus species, TRV infection re-emerged once potatoes were replanted. These findings highlight the remarkable persistence of TRV in agricultural soils, driven by its association with trichodorid nematode vectors and the presence of alternative host plants, including weeds, that maintain virus and vector populations over long periods.
The status of nematodes as virus vectors in South Africa.
Nine nematode species reported from South Africa are implicated in the transmission of plant viruses (Table 1, 2 and 3).The South African trichodorids (members of the nematode family Trichodoridae) can vector Pepper ringspot virus (PepRSV) and Tobacco Rattle Virus (TRV). The South African longidorids (members of the family Longidoridae) vectors the Arabis mosaic virus and Grapevine fanleaf virus. None of the 10 Trichodorus species reported from South Africa are implicated in virus transmission.
Table 1. Summary of key nematode groups, virus associations and crops affected in South Africa.
| Family Trichodoridae | Family Longidoridae | |
| Common name | Stubby-root nematode | Dagger nematode or needle nematode |
| Species reported in SA | Nanidorus minor Paratrichodorus allius Paratrichodorus porosus Paratrichodorus teres, | Paralongidorus maximus Xiphinema americanum Xiphinema diversicaudatum Xiphinema index Xiphinema italiae |
| Viruses transmitted around the world | Tobraviruses Pea early browning virus (PEBV) Pepper ringspot virus (PepRSV) Tobacco rattle virus (TRV) | Nepoviruses ~ 40 species worldwide, for example: Arabis mosaic virus (ArMV) Blueberry leaf mottle virus (BLMoV) Cherry leaf roll virus (CLRV) Grapevine fanleaf virus (GFLV) Peach rosette mosaic virus (PRMV) Raspberry ringspot virus (RpRSV) Tomato black ring virus (TBRV) Tomato ringspot virus (ToRSV) |
| Viruses vectored by nematodes reported in SA | Pepper ringspot virus | Grapevine fanleaf virus Arabis mosaic virus |
| Crops affected | Ornamentals, potatoes, vegetables, weeds | Grapevine, ornamentals, fruit crops |
Table 2. Plant host of South African Trichodoridae species implicated as virus vectors
| Associated plant | Nanidorus minor | Paratrichodorus allius | Paratrichodorus porosus | Paratrichodorus teres |
| Fine Bermuda grass | + | |||
| African mahogany | + | + | ||
| Aloe sp. | + | |||
| Apple | + | + | ||
| Apricot | + | |||
| Artichoke | + | |||
| Arum Lily | + | |||
| Aster sp. | + | |||
| Australin red cedar | + | + | ||
| Avocado | + | + | ||
| Banana | + | + | + | |
| Barley | + | |||
| Beans | + | + | ||
| Beetroot | + | |||
| Bell agapanthus | + | |||
| Bent grass | + | + | ||
| Black wattle | + | + | ||
| Blueberry | + | + | ||
| Bowling green – Cynodon sp. | + | + | ||
| Brinjal | + | |||
| Broccoli | + | |||
| Butterfly pea | + | |||
| Butternut squash | + | |||
| Cabbage | + | + | ||
| Calabash | + | |||
| Cannabis sativa | + | |||
| Cape Gooseberry | + | + | ||
| Carrot | + | + | ||
| Cauliflower | + | |||
| Cherry | + | |||
| Chromolaena | + | |||
| Citrus – lemon | + | + | ||
| Citrus – naartjie | + | + | + | |
| Citrus – orange | + | + | ||
| Citrus- unidentified | + | + | + | |
| Clover | + | |||
| Coffee | + | |||
| Common cluster fig | + | |||
| Common dubbeltjie | + | |||
| Common finger grass | + | |||
| Common reed | + | |||
| Common sillyberry | + | |||
| Cosmos | + | |||
| Cotton | + | + | ||
| Cowpea | + | |||
| Cuban Mahogany | + | |||
| Cucumber | + | |||
| Cynodon sp. | + | |||
| Dainty pavonia | + | |||
| Date palm | + | |||
| Dolichos sp. | + | |||
| Eucalyptus Plantation | + | |||
| Eucalyptus sp. | + | |||
| Fallow | + | |||
| Fever tree | + | |||
| Fig | + | + | ||
| Garlic | + | |||
| Gemsquash | + | |||
| Golf course – Agrostis sp. | + | |||
| Golf Course – Bent grass | + | |||
| Golf Course – Bermuda-Bent Grass mix | + | |||
| Golf Course – Cynodon sp. | + | |||
| Golf Course – Kikuyu | + | |||
| Golf Course – Transvaal quick grass | + | |||
| Grapevine | + | + | ||
| Grenadilla | + | |||
| Groundnut | + | |||
| Guava | + | |||
| Hartebeeskaroo | + | |||
| Hibiscus sp. | + | |||
| Hypericum sp. | + | |||
| Indigenous forest | + | + | ||
| Kale | + | |||
| Kenaf | + | |||
| Khaki Weed | + | |||
| Kikuyu grass | + | |||
| Kiwi fruit | + | |||
| Lablab-bean | + | |||
| Lantana | + | + | ||
| Lavendula sp. | + | |||
| Lawn | + | |||
| Lawn – Kikuyu | + | |||
| Lawn – LM Grass | + | |||
| Lemon balm | + | |||
| Lentils | + | |||
| Lettuce | + | + | ||
| Litchi | + | |||
| Lucerne | + | |||
| Macadamia nut | + | |||
| Maize | + | + | ||
| Mango | + | |||
| Mirror bush | + | |||
| Mung beans | + | |||
| Mustard | + | + | ||
| Natural veld – dune vegetation | + | |||
| Natural veld – ferns | + | |||
| Natural veld – fynbos | + | |||
| Natural veld – grass (unidentified) | + | + | + | + |
| Natural veld – mountain veld | + | + | ||
| Natural veld – unidentified | + | + | + | |
| Natural veld – wetland vegetation | + | |||
| Nectarine | + | + | ||
| Oats – black | + | |||
| Oats – common | + | |||
| Okra | + | + | ||
| Onion | + | + | ||
| Papyrus | + | |||
| Pastures (Unidentified) | + | |||
| Pea | + | |||
| Peach | + | + | + | |
| Pear | + | |||
| Pecan nut | + | + | ||
| Pepper- sweet | + | |||
| Pineapple | + | |||
| Pinus plantation | + | + | ||
| Pinus sp. | + | |||
| Plum | + | + | ||
| Polygonum sp. | + | |||
| Potato | + | + | ||
| Prickley pear | + | |||
| Pumpkin | + | |||
| Purple nutsedge | + | |||
| Purple top | + | |||
| Quercus sp. | + | + | ||
| Quince | + | |||
| Rice | + | |||
| Rocket | + | |||
| Rooibos | + | |||
| Rosa sp. | + | + | ||
| Rye grass | + | |||
| Salix sp. | + | |||
| Shepherd’s tree | + | |||
| Shrubs (unidentified) | + | |||
| Sorghum | + | |||
| Soyabeans | + | + | ||
| Spinach | + | |||
| Sport field – bent grass | + | |||
| Sport field – kikuyu | + | |||
| Sport field – LM Grass | + | |||
| Sport field – perennial rye grass | + | |||
| Stalked bulbine | + | |||
| Sticky everlasting | + | |||
| Strawberry | + | + | ||
| Strawberry clover | + | |||
| Strelitzia sp. | + | |||
| Sugar beet | + | |||
| Sugarcane | + | + | ||
| Sunflower | + | |||
| Sweet potato | + | |||
| Sweet thorn | + | |||
| Tea | + | + | ||
| Tobacco | + | + | ||
| Tomato | + | + | ||
| Walnut | + | + | ||
| Weeping love grass | + | |||
| Weeping willow | + | |||
| Wheat | + | |||
| Wild dagga | + | |||
| Yellow bush | + | + |
Table 3. Plant host of Longidoridae species implicated as virus vectors
| Paralongidorus maximus | Xiphinema americanum | Xiphinema diversicaudatum | Xiphinema index | Xiphinema italiae | |
| Apple | + | ||||
| Apricot | + | ||||
| Arum lily | + | ||||
| Banana | + | ||||
| Beetroot | + | ||||
| Bell agapanthus | + | ||||
| Citrus – unidentified | + | ||||
| Combretum sp. | + | ||||
| Couch grass | + | ||||
| Date palm | + | ||||
| Eucalyptus plantation | + | + | |||
| Eucalyptus sp. | + | ||||
| Fig | + | ||||
| Grapevine | + | + | + | + | |
| Honeybush | + | ||||
| Indigenous forest | + | + | |||
| Lawn | + | ||||
| Lucerne | + | ||||
| Natural veld | + | + | |||
| Natural veld – dune vegatation | + | + | |||
| Natural veld – grass (unidentified) | + | + | |||
| Nectarine | + | ||||
| Peach | + | + | + | ||
| Pear | + | ||||
| Pineapple | + | ||||
| Pinus plantation | + | ||||
| Pinus sp. | + | ||||
| Plum | + | ||||
| Prune | + | ||||
| Purple nutsedge | + | ||||
| Sisal | + | ||||
| Sugarcane | + | + | |||
| Sweet potato | + | ||||
| Tobacco | + | ||||
| Tomato | + | + | |||
| Wheat | + | ||||
| Wild date palm | + |
The South African trichodorids implicated as virus vectors are polyphagous (Table 2) and with a wide distribution in South Africa (Figs 1-4). Nanidorus minor can vector both Pepper Ring Spot Virus (PepRSV) and Tobacco Rattle Virus (TRV). Work done by Dr Lindy Esterhuizen and her team at ARC-PHP proved transmission of PepRSV by N. minor in a series of soil transmission experiments from potatoes and sunflower as virus source to various hosts, including spinach, tomatoes, peppers sunflowers and various grass crops. In addition, the presence of PepRSV in the nematode vector was confirmed by RT-PCR. TRV can be transmitted by both P. allius and P. porosus, and P. teres is implicated as a vector of Pea Early Browning Virus (PEBV), these two viruses have however not been reported in South Africa.

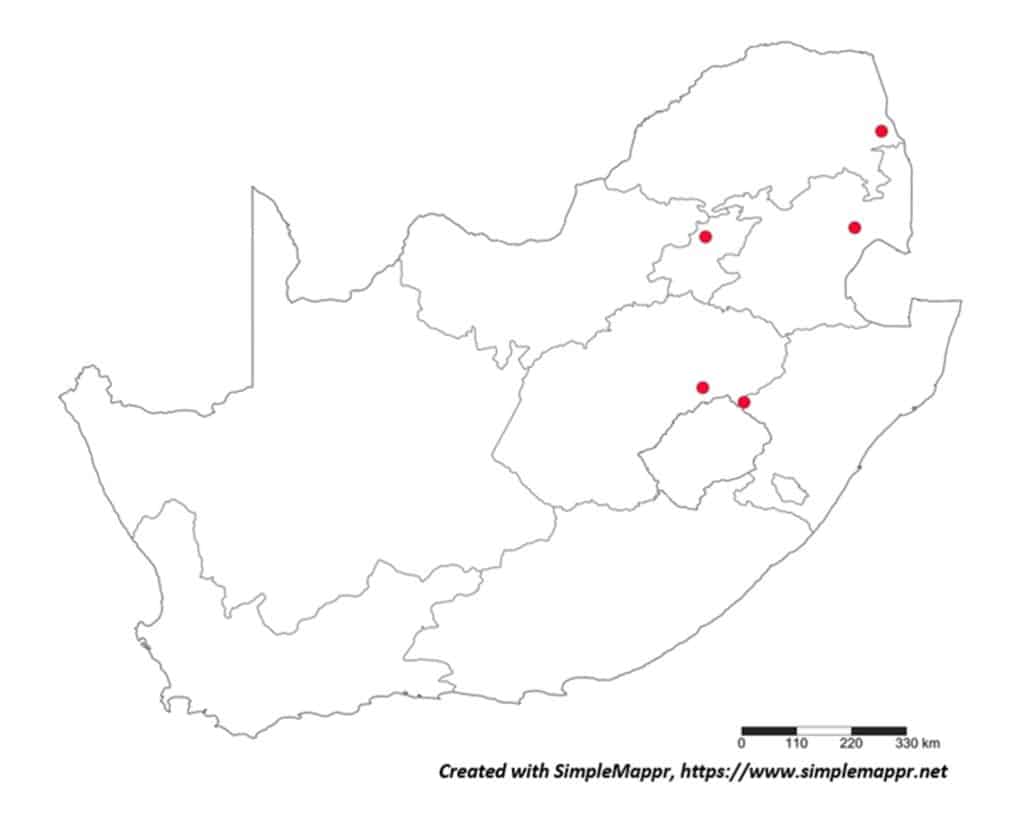
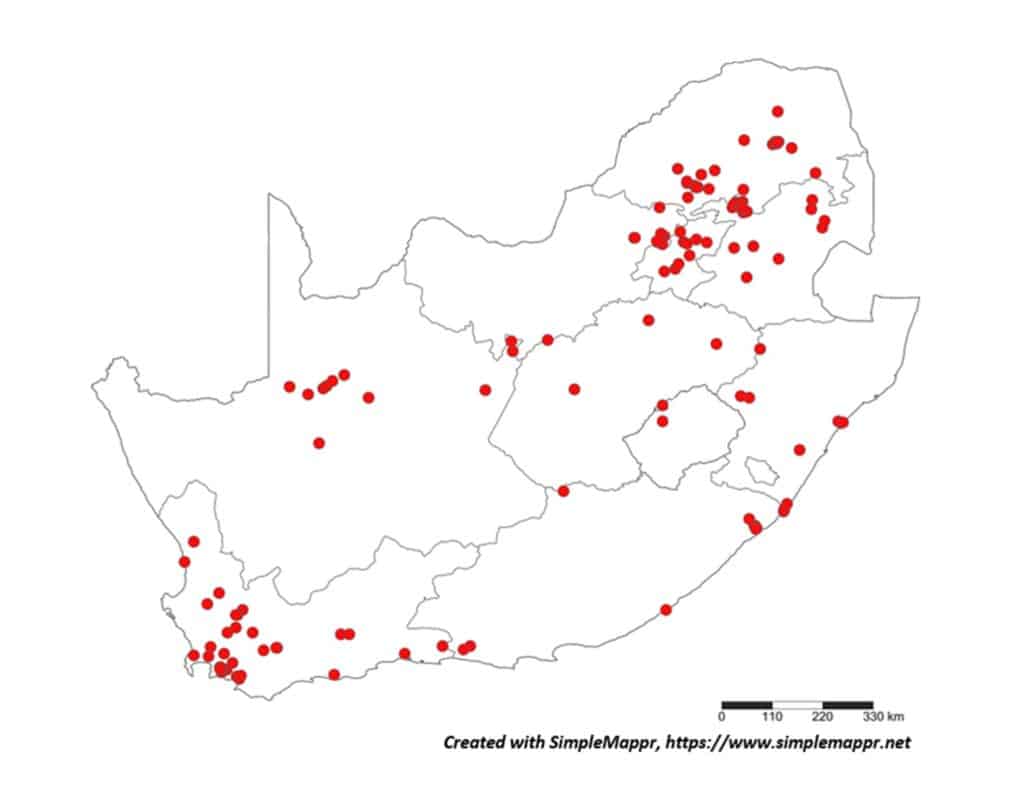
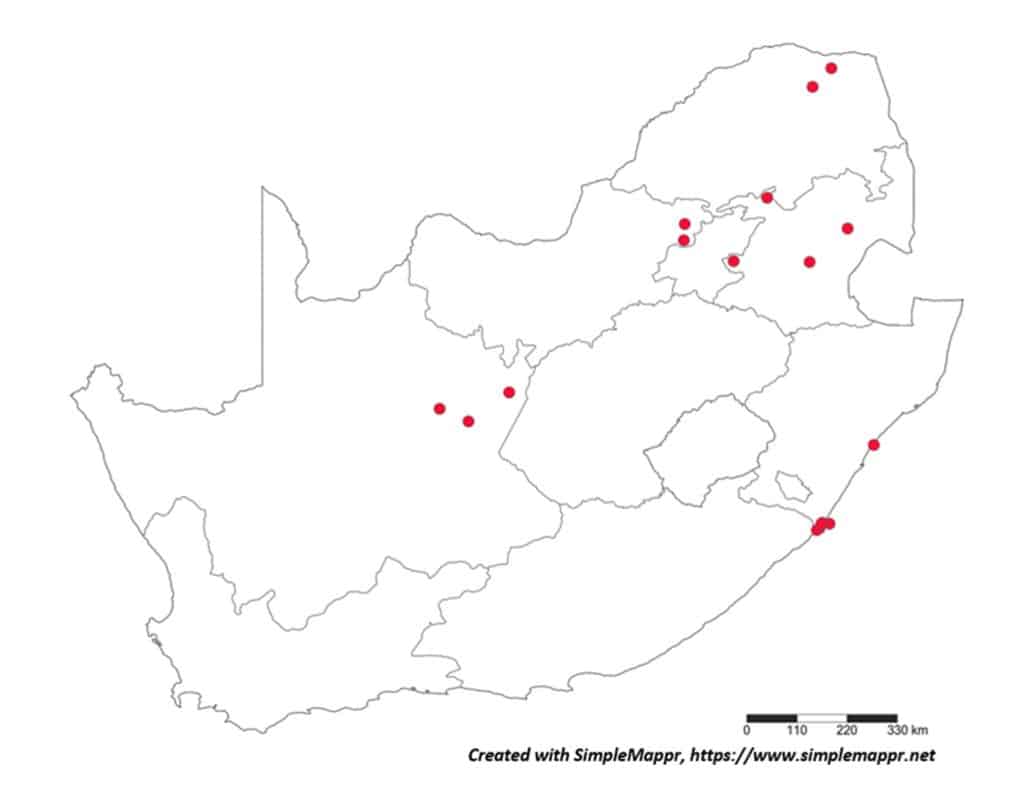
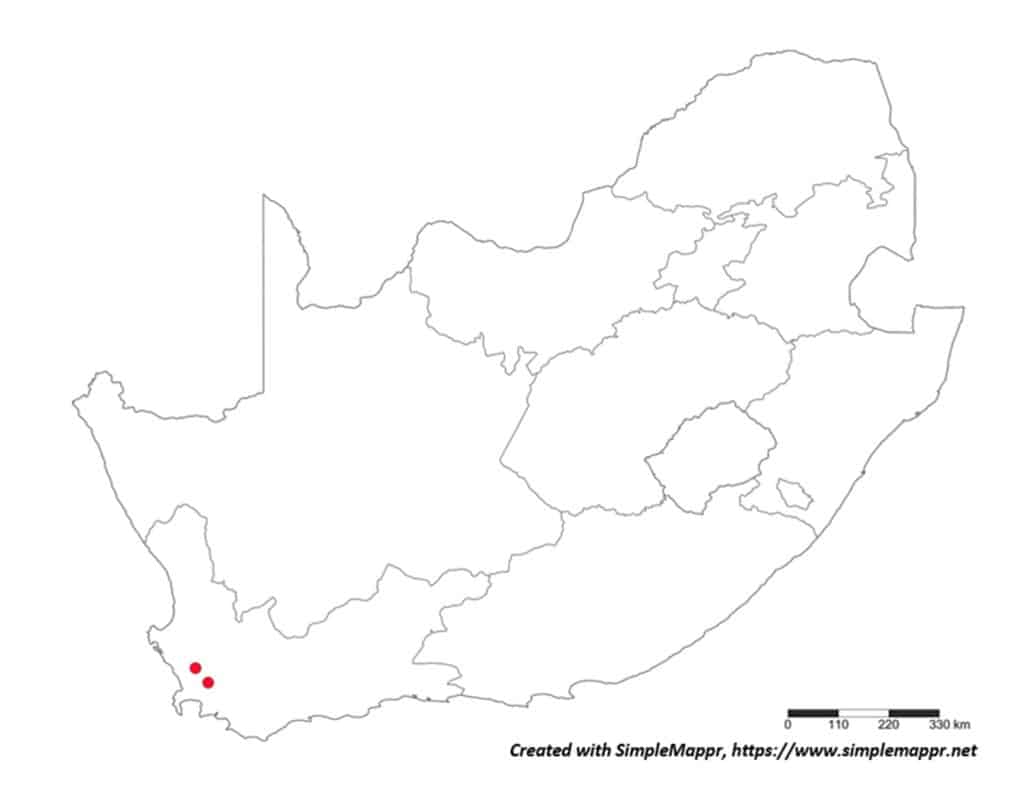
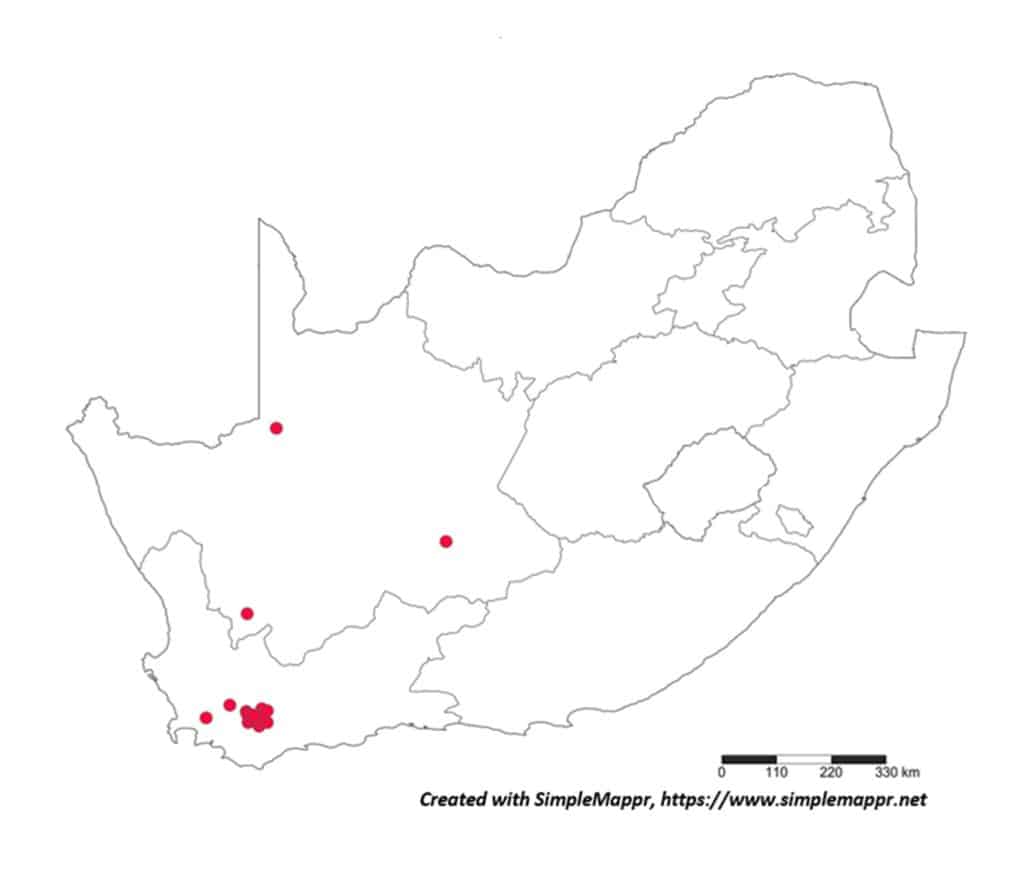
With the primary source of data regarding the distribution of plant-parasitic nematodes in South Africa the datasets regarding both associated plants and distribution was obtained from the South African Plant-Parasitic Nematode Survey (SAPPNS) and the National Collection of Nematodes (NCN). The SAPPNS with now more than 10 100 data locality points was established in 1987 and has documented nematode species across all the biomes over nearly four decades (Marais et al., CHIPS September/October 2023). The National Collection of Nematodes was established in 1961 and house nearly 200 000 nematode specimens. Thus, according to SAPPNS and NCN data, the distribution of Paralongidorus maximus and X. diversicaudatum is restricted to two localities in the Western Cape Province (Fig. 5). In contrast, three Xiphinema species (X. americanum, X. italiae and X. index) show a much more extensive distribution across South Africa (Figs. 6–9). It should be noted that X. index is a regulated pest organism in South Africa (Government Gazette No. 11838, 30 May 2025). Its presence has important phytosanitary implications, as it is an efficient vector of grapevine fanleaf virus and can contribute to the long-term persistence of this disease in vineyards.
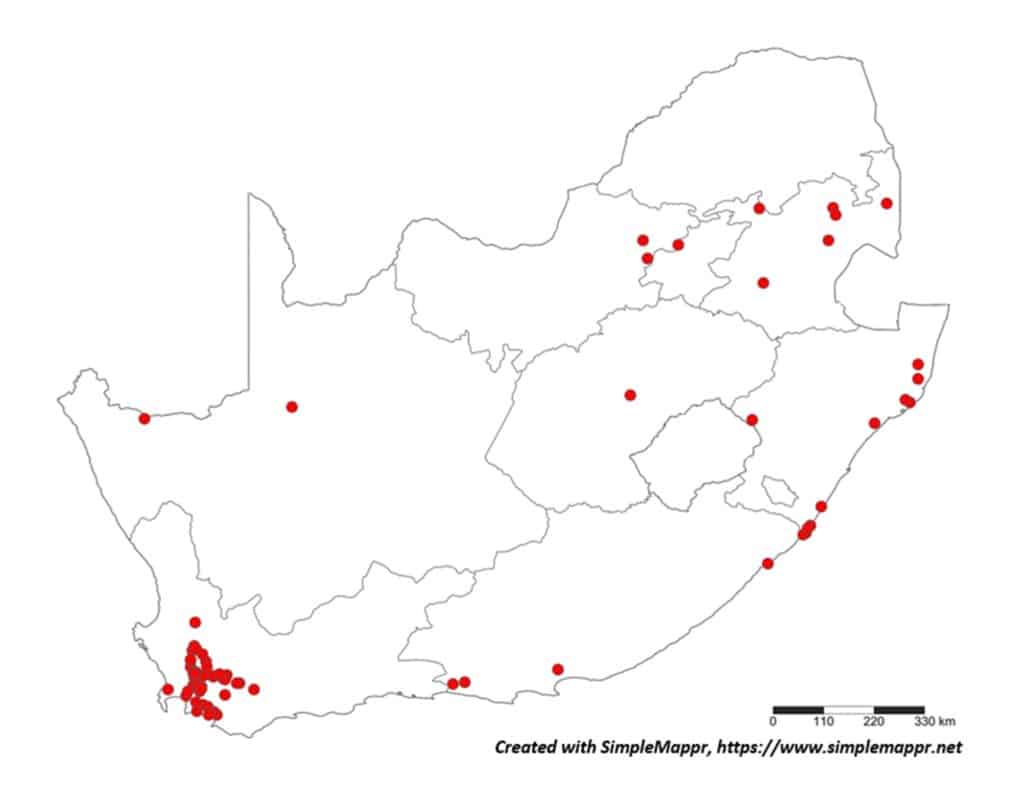


Why are genera of the family Trichodoridae hard to manage?
We can begin answering this question by stating that these nematodes have unique characteristics. Firstly being polyphagous with an extremely diverse host range that includes both monocotyledonous and dicotyledonous plants, thus making crop rotation as a means of nematode control extremely difficult. These nematodes are able to survive in deeper soil layers and can migrate vertically to depths of up to approximately 60–70cm, particularly in lighter soils or in response to unfavourable surface conditions. This vertical movement allows populations to escape some control measures, such as fumigation or nematicide applications, which are often limited to the upper soil layers. The ability to move through soil layers also gave them the ability to quickly move to the rhizosphere of newly planted crops. So, while nematicides can temporarily reduce the trichodorid populations, the nematodes in the deeper soil layers or those associated with weeds can re-infest the rhizosphere of newly planted crops and so making long-term control problematic. Trichodorids thrive in sandy soils but are quite capable to survive in varying soil situation as illustrated by their diverse geographical distribution in South Africa. Limited chemical options and the need for species identification of the nematodes further complicates management. Effective management often requires an integrated approach (IPM) that includes maintaining strict weed control, to eliminate potential host only using certified, virus-free and nematode free seed potatoes, and monitoring for the presence of the nematodes and the virus.
Managing nematode-transmitted viruses starts with knowing what is present in the soil and in the planting material entering a production system. Certification schemes for seed potatoes, grapevine propagation material and fruit trees play a critical role in supplying virus-free planting material and limiting long-distance spread of these pathogens. Field-level decisions can then be strengthened through soil and plant testing for nematode vectors and viruses. In this context, the Agricultural Research Council offers diagnostic support to producers, providing testing capacity for both viruses and nematodes to support informed, system-level management. By managing production systems based on sound knowledge of soil, planting material and crop history, long-term disease risk can be reduced more sustainably. – Mariette Marais & Lindy Esterhuizen, Agricultural Research Council-Plant Health and Protection

